Key Outcomes
Developed an AI-powered TENS device prototype delivering personalized electrical stimulation therapy based on [patient-reported pain levels and treatment response data]
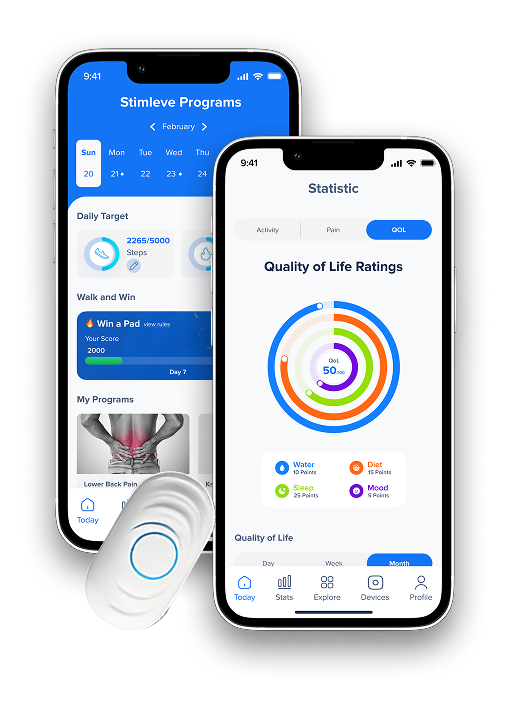
Built a companion mobile app with [BLE device control, therapy session management, and pain tracking analytics]
Designed a [machine learning pipeline] that adapts stimulation parameters (frequency, pulse width, intensity) based on individual patient response patterns
The Challenge
Before partnering with Iottive, [Stimleve] was facing several issues:
One-Size-Fits-All Stimulation
Traditional TENS devices offer fixed programs that don’t adapt to individual pain patterns, body composition, or treatment response [Stimleve] needed a system that personalizes therapy parameters using AI rather than relying on manual user adjustment.
Clinical Validation Requirements
An AI-driven pain therapy device requires [clinical evidence] demonstrating efficacy and safety before regulatory submission. The development pathway needed to support both [FDA clearance milestones] and clinical trial data collection.
Patient Compliance and Engagement
Home-use TENS therapy suffers from low adherence — patients forget sessions, use incorrect electrode placement, or abandon treatment when immediate relief isn’t perceived. The companion app needed to drive consistent therapy usage.
Data-Driven Personalization Pipeline
Adapting stimulation parameters based on patient response requires collecting [pain scores, session data, and outcome metrics] over time, then training models that predict optimal settings for each individual — a complex data science and firmware integration challenge.
Our Solution
Iottive delivered a complete AIoT solution under a full-cycle product development model.
Bluetooth & IoT Expertise
Engineered BLE communication between the TENS device and companion app, enabling real-time parameter adjustment during therapy sessions and post-session data upload for AI model training.
Design Thinking Approach
Designed the patient experience for [chronic pain sufferers], many of whom have limited mobility and dexterity. Large touch targets, simplified session start, and guided electrode placement using [visual instructions] reduce barriers to consistent use.
Project Planning & Execution
Structured development in [clinical-milestone-aligned phases]: prototype for benchtop testing, functional device for [pilot study], data collection firmware for [clinical trial support], and productionready firmware for [regulatory submission].
Functionalities Delivered
BLE device control (start/stop/adjust during session), AI-recommended therapy programs based on pain type and history, pain score tracking before and after sessions, electrode placement guidance, therapy reminder notifications, session history and trend visualization, and [clinician portal] for remote patient monitoring.
Implementation Highlights
Sensors & Hardware
[Multi-channel TENS stimulator] with adjustable frequency (1–150 Hz), pulse width (50–300 μs), and intensity. Electrode impedance monitoring ensures proper skin contact before therapy begins.
Edge & Connectivity
BLE connection to iOS/Android companion app. On-device safety limits enforced in firmware — the app can request parameter changes, but the device independently enforces maximum intensity and session duration limits.
Firmware & Performance Optimization
Real-time waveform generation with smooth parameter transitions during AI driven adjustments. No abrupt intensity changes — all modifications ramp over [500ms] to prevent patient discomfort.
Data Analytics & Visualization
[Machine learning models] trained on aggregated patient response data to predict optimal stimulation parameters for new patients based on pain type, location, and severity. Individual models refine predictions with each session.
Dashboard & UX
Patient app with one-tap session start, real-time intensity visualization, pain score entry (0–10 scale with visual anchors), and therapy progress charts showing pain reduction trends over weeks.
Security & Compliance
[HIPAA-compliant] data handling for patient health information. [FDA 510(k)] pathway considerations built into firmware architecture from day one. Encrypted BLE and cloud communications.
Results & Impact
- [Functional prototype] completed and validated in bench-top testing
- AI personalization engine demonstrated [measurable improvement] in therapy parameter selection versus fixed programs
- Patient app designed for [accessibility compliance], supporting users with limited dexterity and vision
- [Data collection pipeline] established for clinical validation studies
- [Regulatory pathway] mapped with firmware architecture supporting FDA 510(k) documentation requirements
“Iottive brought the firmware and IoT expertise we needed to turn our AI pain therapy concept into a working device. Their team understood that a medical TENS device isn’t a consumer gadget — safety limits must be enforced in hardware, not just software. The BLE integration and companion app they built collect the session data our AI models need while keeping the patient experience simple enough for daily use.”
— Musa Alhakimi , Stimleve
Lessons & Best Practices
Safety Limits in Firmware, Not Software
Medical electrical stimulation devices must enforce maximum intensity and session duration at the firmware level. App-side limits are a convenience; firmware limits are the safety net.
Design for Regulatory from Day One
Retrofitting [FDA documentation requirements] into existing firmware is expensive. Building audit logging, version control, and traceability into the development process from the start saved months of regulatory preparation.
Collect Data for AI Before Deploying AI
The AI personalization engine required [thousands of session records] before it could outperform fixed programs. Phase 1 focused on data collection with manual programs; Phase 2 activated AI recommendations.
Ramp All Parameter Changes
Abrupt stimulation changes cause discomfort and erode patient trust. All intensity, frequency, and pulse width transitions use [500ms ramps], making AI-driven adjustments imperceptible to the patient.

Technology
Comprehensive IoT Technology Stack
From devices and connectivity to cloud, apps, and security — we leverage a full-stack IoT ecosystem to build scalable, secure, and future-ready solutions.
BLE connectivity
iOS Swift

Android Kotlin

Machine learning personalization engine

HIPAA-compliant cloud data platform

FDA 510(k) aligned firmware architecture

Why Iottive’s the Right Partner
- Medical device firmware expertise: Iottive builds [FDA-pathway-aware] embedded systems with safety-critical firmware architecture, audit logging, and regulatory documentation support
- AI-IoT integration: experience connecting edge devices to cloud-based machine learning pipelines, enabling personalized therapy based on accumulated patient data
- Healthcare UX design: building patient-facing apps for populations with accessibility needs, low health literacy, and limited technology experience
