Key Outcomes
Built a medical-grade iOS companion app (Vagal Tones) controlling tVNS stimulation parameters over BLE 5.2 — frequency, waveform, and intensity — across 50,000+ shipped devices
Delivered research-edition capabilities including double-blind study mode, true sham control, and full parameter access, enabling clinical-grade data collection
Implemented secure firmware OTA updates over BLE, allowing Transcranial Ltd to deploy device improvements to the entire installed base without hardware recalls
The Challenge
Before partnering with Iottive, Transcranial Ltd was facing several issues:
Medical-Grade Software Requirements
The Vagal tVNS stimulator operates under IEC 60601-1 and ISO 13485 safety standards. The companion app needed to enforce strict parameter boundaries, handle BLE disconnections gracefully during active stimulation, and maintain audit-ready session logs requirements far beyond a typical consumer BLE app.
Research vs. Consumer Feature Split
Transcranial sells to two distinct audiences: everyday wellness users (tVNS Stimulator at $399, Vagal Oh! at $499) and clinical researchers who need full parameter control, double-blind protocols, and sham stimulation. A single app had to serve both without overwhelming either.
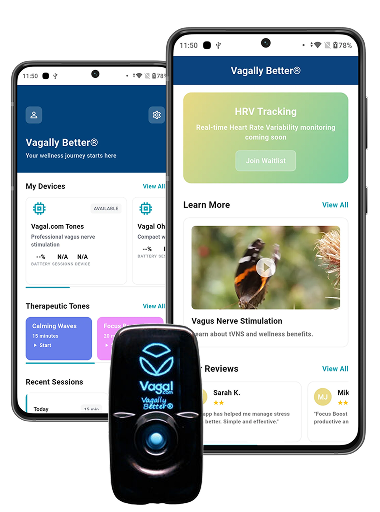
Real-Time Stimulation Control
The device’s EverSteady technology makes 50,000 current adjustments per second. The app had to communicate intensity changes, start/stop commands, and waveform configurations in real time over BLE 5.2 without introducing latency that could affect treatment consistency.
Firmware Lifecycle Management
With 50,000+ units in the field and growing, Transcranial needed a reliable OTA firmware-update pathway. Users range from tech-comfortable early adopters to older wellness consumers — the update process had to be robust and simple.
Our Solution
Iottive delivered a comprehensive BLE companion app under a full-cycle product development model, built to medical-device software standards.
Bluetooth & IoT Expertise
Our engineers designed a BLE 5.2 communication protocol that handles bidirectional data flow: stimulation commands from app to device, and real-time telemetry (battery, session progress, fault alerts) from device to app. The protocol enforces parameter ceilings defined by IEC 60601-1, rejecting out-of-range values at the communication layer before they reach the device.
Design Thinking Approach
We ran iterative design sessions with Transcranial’s clinical and product teams, mapping separate user journeys for Standard Edition (pre-configured protocols, one-tap sessions) and Research Edition (full parameter sliders, study configuration, sham modes). The result: a single app binary with role-based feature access.
Project Planning & Execution
Development followed a milestone-gated model aligned to medical-device documentation requirements. Each sprint produced testable builds with traceability from user requirements to implemented features — supporting Transcranial’s quality-management system under ISO 13485.
Functionalities Delivered
• Standard Edition: pre-configured stimulation protocols, device status, session tracking, firmware updates
• Vagal Oh! support: expanded parameter control for advanced users
• Research Edition: full frequency/waveform/intensity control, double blind study mode, true sham stimulation
• Tone Library: downloadable precision-tuned stimulation protocols deployed to device over BLE
• Session logging with exportable data for clinical and personal use
• Firmware OTA updates with progress tracking and integrity verification
Implementation Highlights
BLE 5.2 Protocol Design
Engineered a custom GATT service set optimized for medical telemetry: separate characteristics for stimulation commands, session state, battery SOC, and firmware transfer. BLE 5.2’s improved throughput and connection stability reduced OTA update times and enabled reliable real-time parameter adjustments.
Safety-First Parameter Control
The app enforces IEC 60601-1 limits at multiple levels: UI sliders are bounded, BLE command payloads are validated before transmission, and the device firmware performs a final check. This triple-layer approach ensures no single software fault can deliver an out-of specification stimulation.
Tone Library & Protocol Management
Built a content-delivery pipeline for Transcranial’s Tone Library — precision-tuned stimulation protocols that users browse, download, and deploy to their device. Each tone includes metadata (target outcome, duration, intensity profile) and is cryptographically signed to prevent tampering.
Research Edition: Double-Blind & Sham
Implemented a study-configuration module where researchers define active and sham arms, assign device codes, and blind both participants and operators. The sham mode delivers sub therapeutic stimulation that mimics the sensation of active treatment, maintaining study integrity.
Firmware OTA Pipeline
Designed a chunked firmware-transfer protocol over BLE 5.2 with CRC verification per block and automatic retry on transmission errors. Users see a progress indicator and the app prevents device use during updates to avoid partial-flash states.
Security & Compliance
All session data is encrypted at rest. BLE pairing uses Secure Connections (LE Secure Connections with LESC). The app architecture supports Transcranial’s ISO 13485 QMS with traceable build artifacts, versioned BLE protocol specifications, and session-audit logs.
Results & Impact
- 50,000+ devices actively paired with Vagal Tones, spanning Standard, Vagal Oh!, and Research editions
- 4.6-star rating across 17,231 verified user reviews — reflecting both app quality and device satisfaction
- Research Edition adopted by clinical teams running double-blind tVNS studies, replacing manual stimulation logging
- Firmware OTA success rate above 99% across the installed base, with zero reported bricked devices
- User-reported outcomes supported by the app’s session data: 25% improvement in focus, 40% reduction in post-meal fatigue, 3× faster sleep onset
“Building a companion app for a medical stimulation device isn’t like building a fitness tracker. You need engineers who understand that a BLE dropout during a session isn’t an annoyance — it’s a safety event. Iottive brought that mindset from day one. Their work on the Research Edition, especially the double-blind and sham features, gave our clinical partners tools they couldn’t find anywhere else.”
— Michael Oxley, Vagal / Vagal Tones
Lessons & Best Practices
Enforce Safety at Every Layer
Medical-device software cannot rely on a single validation point. We implemented parameter bounds in the UI, the BLE command encoder, and the device firmware — so a fault in any one layer is caught by the others.
Design Two Apps in One Binary
Serving both consumers and researchers from a single codebase required disciplined feature flagging and role-based access. This reduced maintenance burden while ensuring each audience sees only what they need.
Treat BLE Disconnection as a Safety Event
We designed the stimulation-control protocol so the device defaults to a safe state (ramp-down and stop) if BLE connectivity is lost mid-session — eliminating the risk of uncontrolled stimulation.
Plan OTA for Scale from the Start
With 50,000+ devices in the field, firmware updates must be reliable at volume. Chunked transfers, per-block CRC, and automatic retry were non-negotiable design decisions, not afterthoughts.

Technology
Comprehensive IoT Technology Stack
From devices and connectivity to cloud, apps, and security — we leverage a full-stack IoT ecosystem to build scalable, secure, and future-ready solutions.
iOS (Swift, SwiftUI)
Bluetooth

GATT Services

DFU

Why Iottive’s the Right Partner
- Medical-device BLE expertise: we understand the difference between a consumer disconnect and a safety event, and we engineer accordingly — from parameter validation to graceful fault handling under IEC 60601-1
- Research-grade feature development: our team has built double-blind study modes, sham controls, and audit-ready session logging — capabilities that clinical research teams require and most app developers have never encountered
- Proven scale: supporting 50,000+ active devices with reliable OTA firmware updates demonstrates our ability to build systems that perform under real-world, high-volume conditions
