Key Outcomes
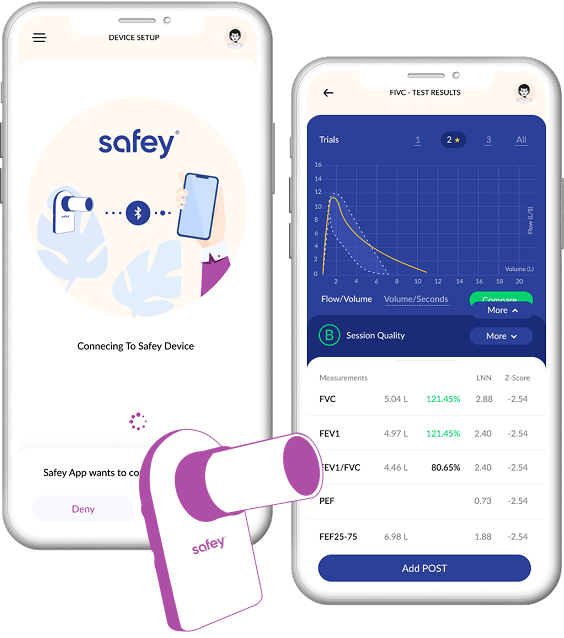
Delivered the companion app for an FDA 510(k) cleared, ATS/ERS-compliant Bluetooth spirometer enabling home-based lung function monitoring for asthma and COPD patients
Implemented real-time BLE data capture for FEV1, FVC, PEF, FEV6, and additional spirometry parameters with clinical-grade accuracy
Enabled remote telehealth monitoring by transmitting spirometry data from patient homes to clinician dashboards, reducing the need for in-clinic pulmonary function tests
The Challenge
Before partnering with Iottive, Safey Medical Devices was facing several issues:
Complex Medical Device Communication
The Safey Pocket Spirometer (95×58×8.2mm) transmits multiple pulmonary function parameters over Bluetooth. Building a companion app that reliably captures every data point during a forced expiratory maneuver — which lasts only a few seconds — required precise BLE timing and error handling.
FDA and Clinical Compliance Requirements
As a companion to an FDA 510(k)-cleared device, the app had to meet stringent requirements for data integrity, traceability, and display accuracy. Any data loss or display error during a spirometry test could compromise clinical decisions.
ATS/ERS Standards Compliance
Spirometry results must comply with American Thoracic Society (ATS) and European Respiratory Society (ERS) standards for acceptability and repeatability. The app needed to apply these validation criteria to each test session.
Patient-Friendly Operation
Spirometry is inherently technique-dependent. Patients performing tests at home — without a technician guiding them — needed clear visual coaching cues and immediate feedback on test quality.
Our Solution
Iottive delivered a complete AIoT solution under a full-cycle product development model.
Bluetooth & IoT Expertise
Built robust BLE communication with the Safey Pocket Spirometer, handling real-time data streaming during forced expiratory maneuvers. The protocol manages device discovery, pairing, data capture, and graceful disconnection — all within the tight timing window of a spirometry test.
Design Thinking Approach
Collaborated with Safey’s clinical and regulatory teams to design a patient-facing interface that guides users through the spirometry technique: deep breath in, blast out, keep going. Visual flow-volume curves provide immediate feedback on test quality.
Project Planning & Execution
Delivered across iOS and Android platforms in parallel, with shared BLE protocol libraries. Regulatory documentation was maintained alongside development to support Safey’s FDA submission process.
Functionalities Delivered
BLE spirometer pairing and data capture, real time flow-volume and volume-time curve display, FEV1/FVC/PEF/FEV6 parameter calculation and display, ATS/ERS test quality validation, historical trend tracking, and remote monitoring data transmission for telehealth workflows.
Implementation Highlights
BLE Spirometry Data Protocol
Implemented a custom BLE data protocol optimized for real-time spirometry data streaming. The app captures high-frequency flow data during the 6–15 second forced expiratory maneuver, buffering and processing data with zero packet loss.
Clinical Parameter Calculation
Built on-device calculation engines for FEV1, FVC, PEF, FEV6, FEV1/FVC ratio, and additional derived parameters. Results are calculated per ATS/ERS standards with appropriate rounding and unit display.
Flow-Volume Curve Rendering
Real-time rendering of flow-volume and volume time curves during the test provides immediate visual feedback. Post-test overlay views allow comparison of multiple efforts within a session.
ATS/ERS Quality Validation
Each test is automatically evaluated against ATS/ERS acceptability criteria (back extrapolated volume, end-of-test plateau, reproducibility). Patients receive clear pass/fail indicators with coaching suggestions for failed attempts.
Trend Analysis & Remote Monitoring
Historical spirometry data is tracked over weeks and months, with trend lines highlighting improvement or decline. Data packages are formatted for transmission to remote clinician dashboards.
Regulatory Documentation Support
Maintained traceability matrices linking app requirements to test protocols and validation results, supporting Safey’s FDA 510(k) submission and ongoing regulatory compliance.
Results & Impact
- Companion app deployed alongside an FDA 510(k)-cleared spirometer — one of the few pocket sized, Bluetooth-connected spirometers on the market
- Accurate capture and display of 5+ spirometry parameters in real-time during forced expiratory maneuvers
- ATS/ERS quality validation enables patients to perform clinically valid tests at home without technician supervision
- Remote monitoring capability reduces the need for in-clinic pulmonary function tests, improving access for rural and mobility-limited patients
- Cross-platform delivery (iOS + Android) maximized patient reach from initial launch
“Building a companion app for an FDA-cleared spirometer is not standard app development. Iottive understood the clinical requirements, the BLE timing constraints, and the regulatory stakes. They delivered an app that our patients trust and our regulators accept.”
— Taher Moiyed, Safey Medical Devices
Lessons & Best Practices
Treat BLE Timing as a Clinical Requirement
In spirometry, a dropped BLE packet during a 6 second forced exhalation can invalidate an entire test. BLE protocol design was driven by clinical requirements, not standard connectivity patterns.
Validate Against Gold-Standard Equipment
App-calculated parameters were validated against hospital-grade spirometers to ensure clinical accuracy before deployment. This validation data supported the FDA submission.
Coach the Patient, Not Just Display Data
Home spirometry succeeds or fails on patient technique. Investing in real-time coaching cues — visual animations and clear pass/fail feedback — had more impact on data quality than any backend improvement.
Maintain Regulatory Traceability from Day One
Retroactively documenting requirements traceability for regulatory submissions is expensive and error prone. Embedding traceability into the development workflow from sprint one saved significant effort during FDA review.

Technology
Comprehensive IoT Technology Stack
From devices and connectivity to cloud, apps, and security — we leverage a full-stack IoT ecosystem to build scalable, secure, and future-ready solutions.
iOS (Swift)
BLE

cloud

Why Iottive’s the Right Partner
- Specialized BLE expertise for medical device communication — including the timing precision required for real-time spirometry data capture
- Experience supporting FDA regulatory processes with development traceability, validation protocols, and compliance documentation
- Cross-platform mobile development (iOS + Android) with shared BLE protocol libraries, ensuring consistent clinical accuracy across devices
